Fig. 1
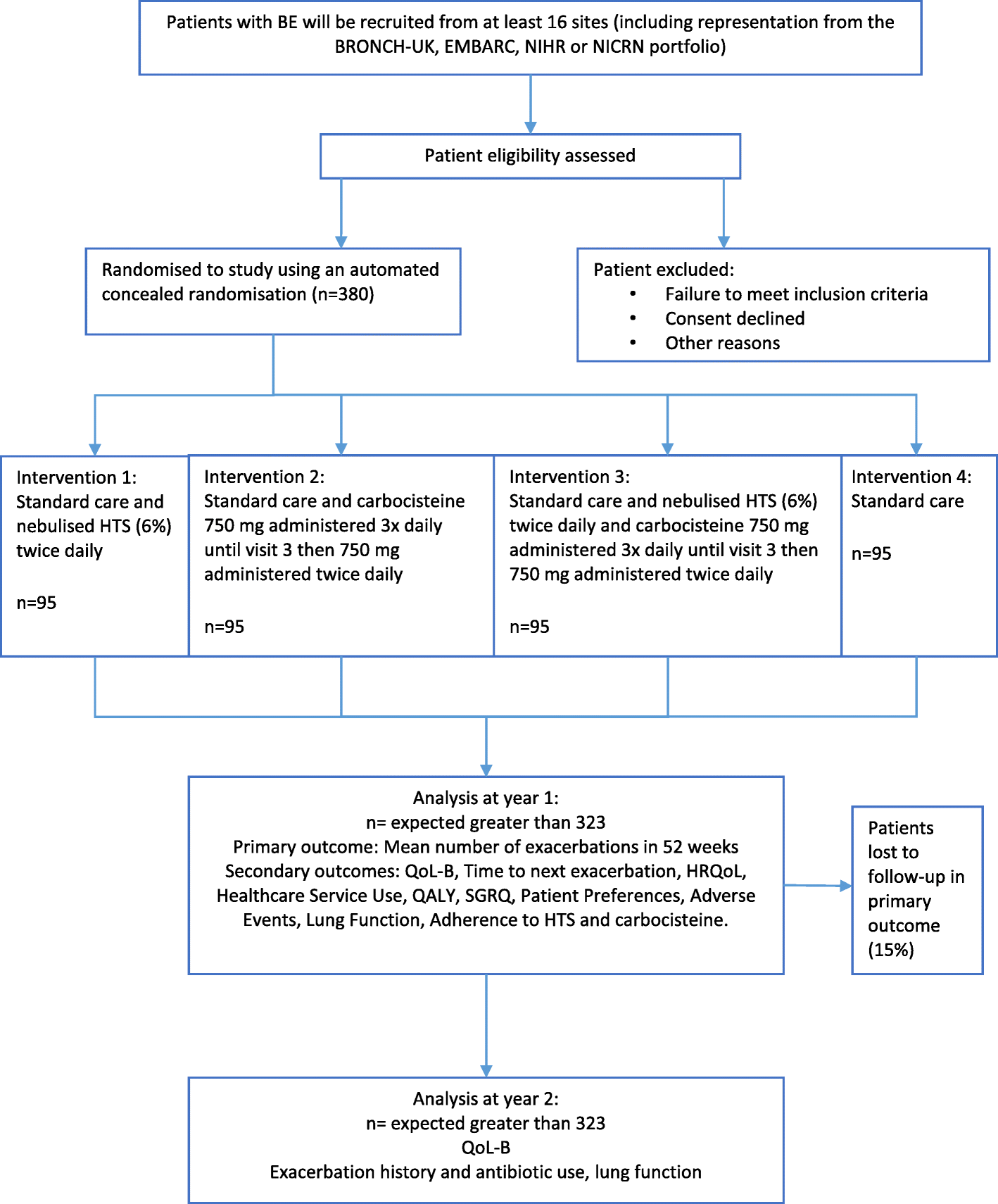
Study schematic. BE, bronchiectasis; BRONCH-UK, UK Bronchiectasis Registry; EMBARC, European Multicentre Bronchiectasis Audit and Research Collaboration; NIHR, National Institute for Health Research; NICRN, Northern Ireland Clinical research Network; HTS, hypertonic saline; QoL-B, Quality of Life - Bronchiectasis; HRQoL, health-related quality of life; QALY, quality-adjusted life year; SGRQ, Saint George’s Respiratory Questionnaire
